
For US Healthcare Professionals

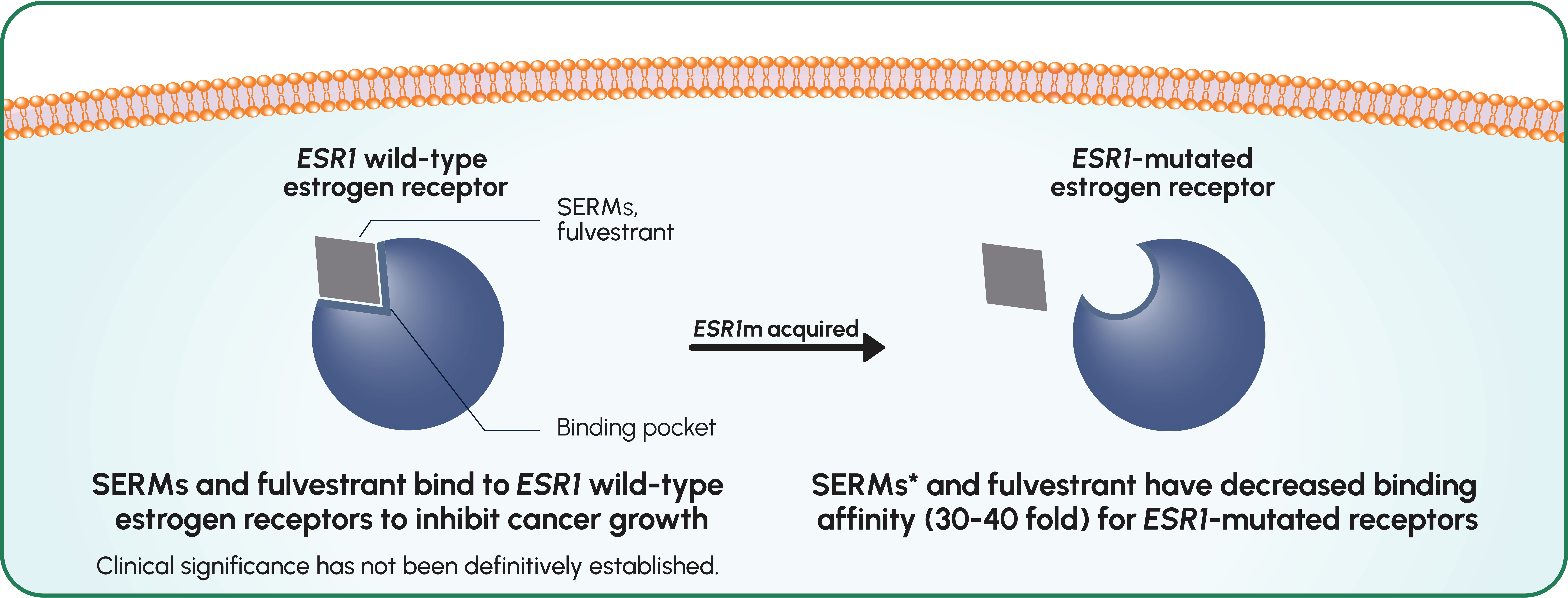
Adapted with permission from Brett JO, Spring LM, Bardia A, Wander SA. ESR1 mutation as an emerging clinical biomarker in metastatic hormone receptor-positive breast cancer. Breast Cancer Res. 2021;23(1):85.
aBC, advanced breast cancer; ER+, estrogen receptor-positive; ESR1, estrogen receptor 1; ESR1m, estrogen receptor 1 mutation; HER2-, human epidermal growth factor receptor 2-negative; mBC, metastatic breast cancer; SERMs, selective estrogen receptor modulators.
*Specifically refers to tamoxifen from this analysis.
-
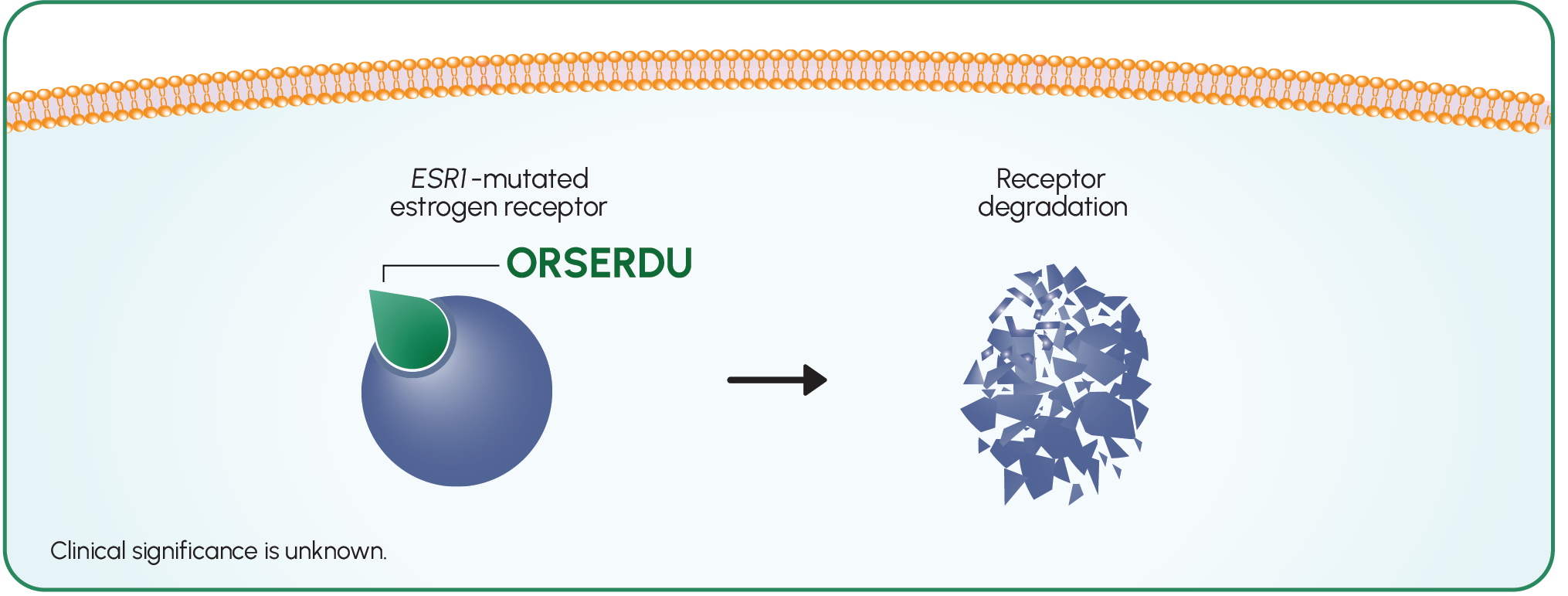
ORSERDU is an ER antagonist that binds to ER-alpha4
-
ORSERDU demonstrated in vitro and in vivo antitumor activity, including in cells resistant to fulvestrant and a CDK4/6i, and cells with ESR1 mutations4
-
ORSERDU inhibited 17β-estradiol-mediated cell proliferation at concentrations inducing degradation of ER-alpha protein mediated through the proteasomal pathway in ER+/HER2- breast cancer cells4
-
ORSERDU does not target the PI3K/AKT/mTOR pathway4,6
aBC, advanced breast cancer; AKT, protein kinase B; CDK4/6i, cyclin-dependent kinase 4/6 inhibitor; ER+, estrogen receptor-positive; ESR1, estrogen receptor 1; ET, endocrine therapy; HER2-, human epidermal growth factor receptor 2-negative; mBC, metastatic breast cancer; mTOR, mammalian target of rapamycin; PI3K, phosphoinositide 3-kinase.
Learn more about Dosing
IMPORTANT SAFETY INFORMATION
Warnings and Precautions
-
Dyslipidemia: Hypercholesterolemia and hypertriglyceridemia occurred in patients taking ORSERDU at an incidence of 30% and 27%, respectively. The incidence of Grade 3 and 4 hypercholesterolemia and hypertriglyceridemia were 0.9% and 2.2%, respectively. Monitor lipid profile prior to starting and periodically while taking ORSERDU.
-
Embryo-Fetal Toxicity: Based on findings in animals and its mechanism of action, ORSERDU can cause fetal harm when administered to a pregnant woman. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ORSERDU and for 1 week after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ORSERDU and for 1 week after the last dose.
Adverse Reactions
-
Serious adverse reactions occurred in 12% of patients who received ORSERDU. Serious adverse reactions in >1% of patients who received ORSERDU were musculoskeletal pain (1.7%) and nausea (1.3%). Fatal adverse reactions occurred in 1.7% of patients who received ORSERDU, including cardiac arrest, septic shock, diverticulitis, and unknown cause (one patient each).
-
The most common adverse reactions (≥10%), including laboratory abnormalities, of ORSERDU were musculoskeletal pain (41%), nausea (35%), increased cholesterol (30%), increased AST (29%), increased triglycerides (27%), fatigue (26%), decreased hemoglobin (26%), vomiting (19%), increased ALT (17%), decreased sodium (16%), increased creatinine (16%), decreased appetite (15%), diarrhea (13%), headache (12%), constipation (12%), abdominal pain (11%), hot flush (11%), and dyspepsia (10%).
Drug Interactions
-
Concomitant use with CYP3A4 inducers and/or inhibitors: Avoid concomitant use of strong or moderate CYP3A4 inhibitors with ORSERDU. Avoid concomitant use of strong or moderate CYP3A4 inducers with ORSERDU.
Use in Specific Populations
-
Lactation: Advise lactating women to not breastfeed during treatment with ORSERDU and for 1 week after the last dose.
-
Hepatic Impairment: Avoid use of ORSERDU in patients with severe hepatic impairment (Child-Pugh C). Reduce the dose of ORSERDU in patients with moderate hepatic impairment (Child-Pugh B).
The safety and effectiveness of ORSERDU in pediatric patients have not been established.
ORSERDU is available as 345 mg tablets and 86 mg tablets.
INDICATION
ORSERDU (elacestrant) is indicated for the treatment of postmenopausal women or adult men with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, ESR1-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy.
To report SUSPECTED ADVERSE REACTIONS, contact Stemline Therapeutics, Inc. at 1-877-332-7961 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Please see full Prescribing Information.
References: 1. Brett JO, Spring LM, Bardia A, Wander SA. ESR1 mutation as an emerging clinical biomarker in metastatic hormone receptor-positive breast cancer. Breast Cancer Res. 2021;23(1):85. 2. Bihani T, Patel HK, Arlt H, et al. Elacestrant (RAD1901), a selective estrogen receptor degrader (SERD), has antitumor activity in multiple ER+ breast cancer patient-derived xenograft models. Clin Cancer Res. 2017;23(16):4793-4804. 3. Bihani T, Patel HK, Arlt H, et al. Elacestrant (RAD1901), a selective estrogen receptor degrader (SERD), has antitumor activity in multiple ER+ breast cancer patient-derived xenograft models. Clin Cancer Res. 2017;23(16):4793-4804 [supplementary appendix]. 4. ORSERDU [prescribing information]. New York, NY: Stemline Therapeutics, Inc., a Menarini Group Company, 2023. 5. Bardia A, Aftimos P, Bihani T, et al. EMERALD: phase III trial of elacestrant (RAD1901) vs endocrine therapy for previously treated ER+ advanced breast cancer. Future Oncol. 2019;15(28):3209-3218. 6. Patel HK, Bihani T. Selective estrogen receptor modulators (SERMs) and selective estrogen receptor degraders (SERDs) in cancer treatment. Pharmacol Ther. 2018;186:1-24.
This site is intended only for US healthcare professionals. The products discussed in this site may have different product labeling in different countries. The information provided is for educational purposes only.
ORSERDU is a registered trademark of the Menarini Group.
© 2026 Stemline Therapeutics, Inc., a Menarini Group Company. All rights reserved. 03/26 MAT-US-ELA-01221
|
IMPORTANT SAFETY INFORMATION
+
Warnings and Precautions
-
Dyslipidemia: Hypercholesterolemia and hypertriglyceridemia occurred in patients taking ORSERDU at an incidence of 30% and 27%, respectively. The incidence of Grade 3 and 4 hypercholesterolemia and hypertriglyceridemia were 0.9% and 2.2%, respectively. Monitor lipid profile prior to starting and periodically while taking ORSERDU.
-
Embryo-Fetal Toxicity: Based on findings in animals and its mechanism of action, ORSERDU can cause fetal harm when administered to a pregnant woman. Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ORSERDU and for 1 week after the last dose. Advise male patients with female partners of reproductive potential to use effective contraception during treatment with ORSERDU and for 1 week after the last dose.
Adverse Reactions
-
Serious adverse reactions occurred in 12% of patients who received ORSERDU. Serious adverse reactions in >1% of patients who received ORSERDU were musculoskeletal pain (1.7%) and nausea (1.3%). Fatal adverse reactions occurred in 1.7% of patients who received ORSERDU, including cardiac arrest, septic shock, diverticulitis, and unknown cause (one patient each).
-
The most common adverse reactions (≥10%), including laboratory abnormalities, of ORSERDU were musculoskeletal pain (41%), nausea (35%), increased cholesterol (30%), increased AST (29%), increased triglycerides (27%), fatigue (26%), decreased hemoglobin (26%), vomiting (19%), increased ALT (17%), decreased sodium (16%), increased creatinine (16%), decreased appetite (15%), diarrhea (13%), headache (12%), constipation (12%), abdominal pain (11%), hot flush (11%), and dyspepsia (10%).
Drug Interactions
-
Concomitant use with CYP3A4 inducers and/or inhibitors: Avoid concomitant use of strong or moderate CYP3A4 inhibitors with ORSERDU. Avoid concomitant use of strong or moderate CYP3A4 inducers with ORSERDU.
Use in Specific Populations
-
Lactation: Advise lactating women to not breastfeed during treatment with ORSERDU and for 1 week after the last dose.
-
Hepatic Impairment: Avoid use of ORSERDU in patients with severe hepatic impairment (Child-Pugh C). Reduce the dose of ORSERDU in patients with moderate hepatic impairment (Child-Pugh B).
The safety and effectiveness of ORSERDU in pediatric patients have not been established.
ORSERDU is available as 345 mg tablets and 86 mg tablets.
INDICATION
ORSERDU (elacestrant) is indicated for the treatment of postmenopausal women or adult men with estrogen receptor (ER)-positive, human epidermal growth factor receptor 2 (HER2)-negative, ESR1-mutated advanced or metastatic breast cancer with disease progression following at least one line of endocrine therapy.
To report SUSPECTED ADVERSE REACTIONS, contact Stemline Therapeutics, Inc. at 1-877-332-7961 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Please see full Prescribing Information.
References: 1. Brett JO, Spring LM, Bardia A, Wander SA. ESR1 mutation as an emerging clinical biomarker in metastatic hormone receptor-positive breast cancer. Breast Cancer Res. 2021;23(1):85. 2. Bihani T, Patel HK, Arlt H, et al. Elacestrant (RAD1901), a selective estrogen receptor degrader (SERD), has antitumor activity in multiple ER+ breast cancer patient-derived xenograft models. Clin Cancer Res. 2017;23(16):4793-4804. 3. Bihani T, Patel HK, Arlt H, et al. Elacestrant (RAD1901), a selective estrogen receptor degrader (SERD), has antitumor activity in multiple ER+ breast cancer patient-derived xenograft models. Clin Cancer Res. 2017;23(16):4793-4804 [supplementary appendix]. 4. ORSERDU [prescribing information]. New York, NY: Stemline Therapeutics, Inc., a Menarini Group Company, 2023. 5. Bardia A, Aftimos P, Bihani T, et al. EMERALD: phase III trial of elacestrant (RAD1901) vs endocrine therapy for previously treated ER+ advanced breast cancer. Future Oncol. 2019;15(28):3209-3218. 6. Patel HK, Bihani T. Selective estrogen receptor modulators (SERMs) and selective estrogen receptor degraders (SERDs) in cancer treatment. Pharmacol Ther. 2018;186:1-24.

